Black mulberry (Morus nigra Linn.) is one of the important medicinal plants of the Moraceae genus Morus, mainly distributed in Xinjiang. Black mulberry fruit can be used both medicinally and edible. Black mulberry is slightly cold in nature, sweet and sour in taste, enters the liver, kidney, and heart meridians, and is a good fruit for nourishing the mind, nourishing and strengthening. It has the functions of promoting body fluid and quenching thirst, moistening the intestines, nourishing blood and nourishing yin. Regular consumption of black mulberries can improve eyesight and relieve the symptoms of eye fatigue and dryness. Black mulberries are rich in a variety of active substances, including flavonoids, alkaloids, polyphenols, and benzofurans. Modern pharmacological studies have shown that black mulberry has anti-tumor, antioxidant, anti-inflammatory, anti-viral, antihypertensive and hypoglycemic effects.
Metabolic reprogramming can promote the growth and proliferation of cancer cells. Serine metabolism plays a crucial role in tumorigenesis, and increased serine synthesis activity accelerates redox homeostasis, amino acid transport, and nucleotide synthesis, providing cancer cells with a large amount of energy and intermediate metabolites. 3-phosphoglycerate dehydrogenase (PHGDH) is the first rate-limiting enzyme for de novo synthesis of serine, and catalyzes the synthesis of phosphohydroxypyruvate (3-PPyr) from 3-phosphoglycerate (3-PG). Studies have shown that PHGDH is highly expressed in a variety of human tumors, such as melanoma, pancreatic cancer, cervical cancer, and triple-negative breast cancer. Inhibition of PHGDH reduces serine synthesis and inhibits tumor cell proliferation, migration and invasion. Therefore, PHGDH inhibitors may serve as an effective strategy for the treatment of cancer.
In this paper, the fruit of black mulberry was studied, and 20 compounds were isolated, including two new compounds, moranigrineA and morusamine. Among them, MoranigrineA is a rare compound with pyrrole-2-aldehyde and 2-arylbenzofuran structural units, and the authors speculate its biosynthetic pathway. The article evaluated the inhibitory effect of all the isolated compounds on PHGDH, and found that methyl caffeate showed a significant inhibitory effect on PHGDH. Kinetic analysis of enzyme inhibition showed that methyl caffeate was a non-competitive inhibitor relative to the substrate 3-PG and inhibited in a concentration-dependent manner. MST analysis further confirmed the direct interaction of methyl caffeate with PHGDH.
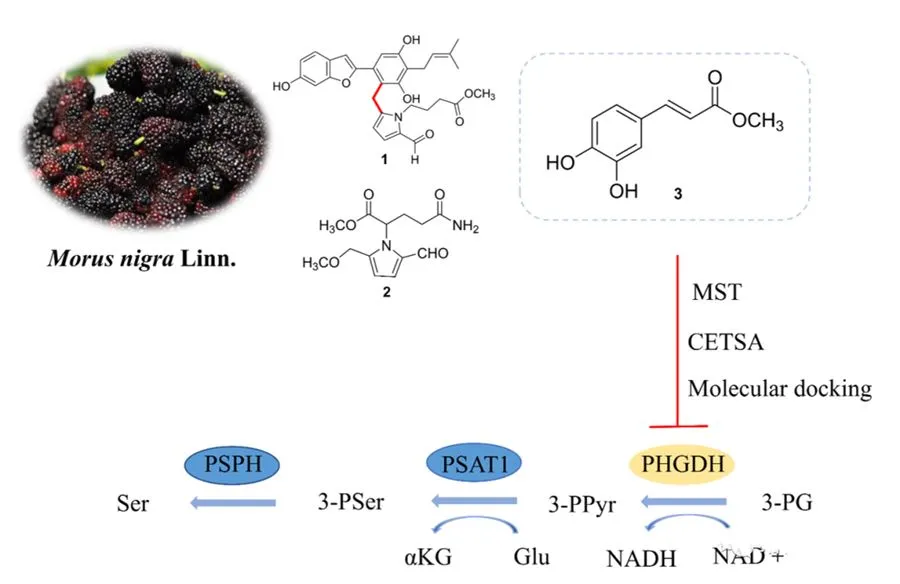
In addition, molecular docking indicated that methyl caffeate binds to the allosteric pocket of PHGDH rather than its active site, which is consistent with the data on enzyme kinetics. Cell experiments showed that methyl caffeate can specifically inhibit the proliferation of HeLa cells; the authors further found that methyl caffeate can promote the apoptosis of HeLa cells and arrest the cell cycle. CETSA and siRNA experiments also proved that methyl caffeate directly targets PHGDH. At the same time, methyl caffeate has obvious inhibitory effect on tumor growth in HeLa cell xenograft mouse model with low toxicity. The above results indicate that methyl caffeate can be used as a natural PHGDH inhibitor, providing a leading compound for the research of antitumor drugs targeting PHGDH, and Mnigra can be used as a functional food for the prevention and adjuvant treatment of cancer.



